Sample Solution
PART-(A)
- Discuss the effect of pressure on the solubility of gases. How is Henry’s law constant determined?
- Draw and explain the mutual solubility curve of nicotine-water system. Which other system exhibits the similar curve?
- (a) What are conjugate solutions? Give one example.
(b) Define metastable equilibrium and differentiate it from true equilibrium - Define any five of the following terms :
(i) A component
(ii) Mole fraction
(iii) Incongruent melting compounds
(iv) Liquid junction potential
(v) Ionic mobility
(vi) Conductometric titration - (a) Give the mathematical expression for Gibbs’ phase rule and calculate the maximum number of phases that can coexist for a one-component system.
(b) Draw the phase diagram for sulphur system and label the sublimation and fusion curves for monoclinic sulphur on it. - (a) How does
muvs \mu \mathrm{vs}
(b) Two substances, X and Y form a simple eutectic system. The melting point of X is more than that ofY Y X X Y Y 40% 40 \% X X - (a) Explain the concentration dependence of molar conductivity for a strong electrolyte
(b) The conductivity of a saturated solution of AgCl at 298 K was found to be 3.41 xx10^(-6)Scm^(-) 3.41 \times 10^{-6} \mathrm{~S} \mathrm{~cm}^{-} ^(1) { }^1 1.6 xx10^(-6)Scm^(-1) 1.6 \times 10^{-6} \mathrm{~S} \mathrm{~cm}^{-1} 138.2Scm^(-1)mol^(-1) 138.2 \mathrm{~S} \mathrm{~cm}^{-1} \mathrm{~mol}^{-1}
8. (a) Describe the moving boundary method for the determination of transference numbers.
(b) In the determination of transference numbers of HCl by moving boundary method a 0.10 M solution of HCl was taken in a cell having uniform area of cross section of1.0cm^(2) 1.0 \mathrm{~cm}^2
8. (a) Describe the moving boundary method for the determination of transference numbers.
(b) In the determination of transference numbers of HCl by moving boundary method a 0.10 M solution of HCl was taken in a cell having uniform area of cross section of
- (a) Outline the requirements for the construction of concentration cells without transference.
(b) The cell reaction for a galvanic cell is given below:
(i) Write the half-cell reactions at the anode and the cathode.
(ii) Calculate the value of cell emf under standard conditions by using the data from Table 7.1.
(iii) Will the reaction be spontaneous as written?
10. (a) Define electrolysis and discuss any one application of electrolysis.
(b) Calculate the time required to get 5 g of magnesium by passing a current of 0.6 A through molten magnesium chloride. (Given:Mm(Mg)=24.25gmol^(-1) \mathrm{Mm}(\mathrm{Mg})=24.25 \mathrm{~g} \mathrm{~mol}^{-1}
11. How will you prepare 2-methylpentanoic acid starting from a suitable alkyl halide? Write the sequence of reactions involved.
12. (a) Which alkyl halide will you use if pentanoic acid is to be obtained? Give the scheme of synthesis involved.
(b) Write the mechanism of amide formation using ethanoic acid and ammonia.
13. (a) What is transesterification? Briefly explain using a suitable example.
(b) Give the products formed when propyl butanoate is reduced using Bouveault-Blanc reduction. Which other reagent can be used to obtain the same products?
14. (a) Using suitable diagram, explain the interconversion of enantiomers of N -methylethanamine.
(b) Explain whyC-N \mathrm{C}-\mathrm{N}
15. (a) Which one is more stable-alkyl diazonium salt or an aryl diazonium salt? Explain.
(b) How can you obtain 4-methylbiphenyl from 4-methylbenzenaine? Write the reactions involved.
16. (a) Explain the Zwitter ionic nature and isoelectric point of a 2-amino acid.
(b) Discuss the chemistry of ninhydrin reaction of 2-amino acids and its importance
17. Discuss the N-protection and deprotection of amino acids using phathaloyl group.
18. Explain sanger method for the N -terminal identification of peptides giving suitable reactions.
19. What are reducing sugars? Which tests are exhibited by them? Give the chemistry of these tests.
20. How was structure of lactose arrived at? Explain. Also write the structure of itsalpha \alpha
(ii) Calculate the value of cell emf under standard conditions by using the data from Table 7.1.
(iii) Will the reaction be spontaneous as written?
10. (a) Define electrolysis and discuss any one application of electrolysis.
(b) Calculate the time required to get 5 g of magnesium by passing a current of 0.6 A through molten magnesium chloride. (Given:
11. How will you prepare 2-methylpentanoic acid starting from a suitable alkyl halide? Write the sequence of reactions involved.
12. (a) Which alkyl halide will you use if pentanoic acid is to be obtained? Give the scheme of synthesis involved.
(b) Write the mechanism of amide formation using ethanoic acid and ammonia.
13. (a) What is transesterification? Briefly explain using a suitable example.
(b) Give the products formed when propyl butanoate is reduced using Bouveault-Blanc reduction. Which other reagent can be used to obtain the same products?
14. (a) Using suitable diagram, explain the interconversion of enantiomers of N -methylethanamine.
(b) Explain why
15. (a) Which one is more stable-alkyl diazonium salt or an aryl diazonium salt? Explain.
(b) How can you obtain 4-methylbiphenyl from 4-methylbenzenaine? Write the reactions involved.
16. (a) Explain the Zwitter ionic nature and isoelectric point of a 2-amino acid.
(b) Discuss the chemistry of ninhydrin reaction of 2-amino acids and its importance
17. Discuss the N-protection and deprotection of amino acids using phathaloyl group.
18. Explain sanger method for the N -terminal identification of peptides giving suitable reactions.
19. What are reducing sugars? Which tests are exhibited by them? Give the chemistry of these tests.
20. How was structure of lactose arrived at? Explain. Also write the structure of its
Answer:
Question:-1
Discuss the effect of pressure on the solubility of gases. How is Henry’s law constant determined?
Answer:
The solubility of gases in liquids is significantly affected by pressure, which is not typically the case for solids dissolving in liquids. While the volume changes of solids in liquids are relatively small and do not influence solubility greatly, gases behave differently due to the much larger compressibility of gases.
In 1803, the English chemist William Henry observed that the solubility of a gas in a liquid increases as the gas pressure is increased at a constant temperature. This relationship is encapsulated in Henry’s Law, which can be stated as:
"At a constant temperature, the partial pressure of a gas over a solution is directly proportional to the concentration of the gas in the solution."
This means that if the partial pressure of the gas above the liquid increases, more gas molecules dissolve in the liquid. For instance, if the pressure of oxygen over a solution is doubled, the amount of oxygen that dissolves in the liquid will also double, provided the temperature remains constant.
Mathematical Expression of Henry’s Law:
Henry’s Law can be mathematically expressed as:
Where:
p p x x k_(H) k_H
This law assumes an ideal solution where the solubility of the gas is directly proportional to its pressure.
Determining the Henry’s Law Constant:
The Henry’s law constant (k_(H) k_H
- Experimental Approach:
- A series of experiments are conducted where the solubility of the gas is measured at different known pressures.
- For each pressure value, the corresponding solubility
x x
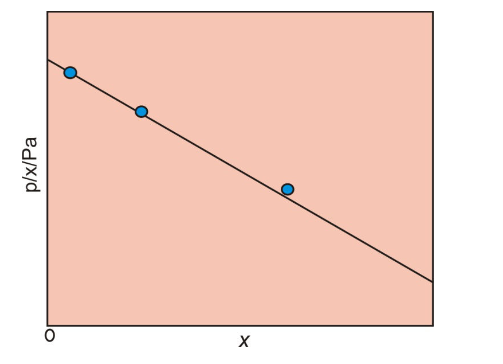
- Plotting the Data:
- A plot is made with the partial pressure
p p x x - According to Henry’s law, this plot should yield a straight line, and the slope of the line gives the value of
k_(H) k_H
- A plot is made with the partial pressure
- Extrapolation:
- The Henry’s law constant is determined by extrapolating the graph to
x=0 x = 0 p//x p/x
- The Henry’s law constant is determined by extrapolating the graph to
- Mathematical Expression of
k_(H) k_H
From the equationp=k_(H)*x p = k_H \cdot x
where p p x x
Example:
Consider a scenario where a gas is dissolved in a solvent under a certain pressure. By increasing the pressure and measuring the solubility, we can determine how much more gas will dissolve. This relationship helps explain phenomena like carbonation in soft drinks—when the bottle is sealed, carbon dioxide is under high pressure, and the gas remains dissolved. Upon opening the bottle, the pressure drops, causing CO₂ to escape from the liquid, reducing its solubility.
Non-Ideal Solutions:
It’s important to note that Henry’s law holds accurately for ideal solutions. In real (non-ideal) solutions, deviations from the ideal behavior can occur, and the Henry’s law constant may vary with changes in concentration. In such cases, the constant is often determined experimentally by extrapolating the graph of p//x p/x x x x=0 x = 0
Graph of p//x p/x x x
The graph is typically linear at lower concentrations and can deviate from linearity at higher concentrations due to non-ideal interactions between the gas and solvent molecules.
The graph is typically linear at lower concentrations and can deviate from linearity at higher concentrations due to non-ideal interactions between the gas and solvent molecules.
Question:-2
Draw and explain the mutual solubility curve of nicotine-water system. Which other system exhibits the similar curve?
Answer:
The mutual solubility curve represents the relationship between the temperature and the solubility of two substances in each other. In the case of a nicotine-water system, this curve illustrates how the solubility of nicotine in water and the solubility of water in nicotine change with temperature.
Mutual Solubility Curve for Nicotine-Water System:
In a nicotine-water system, the solubility of nicotine in water and the solubility of water in nicotine varies as the temperature changes. The curve typically shows the following behavior:
- At Low Temperatures:
At lower temperatures, both nicotine and water have relatively low mutual solubility. The water cannot dissolve much nicotine, and nicotine is not highly soluble in water either. - At High Temperatures:
As the temperature increases, the solubility of both water in nicotine and nicotine in water increases. Both components dissolve more readily as the temperature rises. - Critical Point (Cloud Point):
The curve has a characteristic "cloud point," which is the temperature at which both substances start to form a homogeneous solution. Below this temperature, they separate into two immiscible phases. Above this point, they become completely miscible in each other. - Shape of the Curve:
The mutual solubility curve typically has a concave shape where the solubility of both substances increases with increasing temperature. There’s a point where both substances are completely miscible at higher temperatures and separate at lower temperatures.
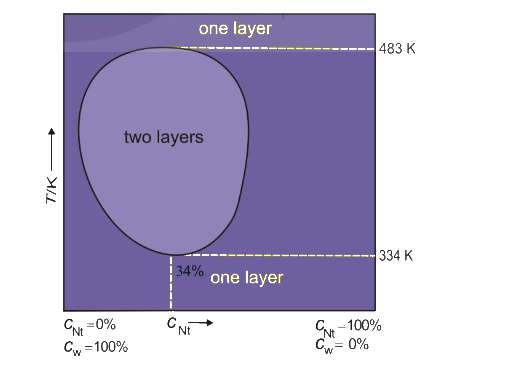
Explanation:
- The mutual solubility of nicotine and water is temperature-dependent. Below a certain temperature, nicotine and water are immiscible, meaning they do not form a homogeneous solution and tend to separate into two phases (a nicotine-rich phase and a water-rich phase).
- As the temperature increases, the solubility of each substance in the other increases, and above a certain temperature, the system becomes completely miscible, forming a single homogeneous solution.
- The critical temperature is the temperature beyond which the two substances are completely miscible and no phase separation occurs.
Similar Systems:
The nicotine-water system is an example of a miscibility gap, where two substances are only partially soluble in each other over a certain temperature range. Another system that exhibits a similar mutual solubility curve is the phenol-water system.
- Phenol-Water System:
Like nicotine and water, phenol and water are partially miscible at lower temperatures but become completely miscible at higher temperatures. The mutual solubility curve for phenol and water also shows a critical temperature point at which they become fully miscible. Below this point, phenol and water separate into two phases, just like the nicotine-water system.
Thus, the nicotine-water system behaves similarly to the phenol-water system and other systems exhibiting partial miscibility that have a concave solubility curve with a critical temperature above which the substances become completely miscible.
Question:-3(a)
What are conjugate solutions? Give one example.
Answer:
Conjugate solutions refer to two solutions of different compositions that can form a pair of opposites in terms of solubility, often referring to the combination of a weak acid and its conjugate base or a weak base and its conjugate acid in a solution. In the context of colloidal solutions, conjugate solutions are typically two-phase systems where one component is more soluble in a specific solvent than the other. These solutions usually show a complementary solubility relationship.
In general, the term conjugate solutions can be applied to systems where a weak acid and its conjugate base (or vice versa) coexist in equilibrium.
Example of Conjugate Solutions:
A common example of conjugate solutions is the acetic acid and acetate ion system. This system consists of:
- Acetic acid (CH₃COOH) – a weak acid that partially dissociates in water.
- Acetate ion (CH₃COO⁻) – the conjugate base of acetic acid.
In solution, acetic acid dissociates partially to produce H⁺ ions and acetate ions. The concentration of acetic acid and acetate ions in equilibrium defines the pH of the solution, and this equilibrium relationship is crucial in buffering solutions.
The conjugate acid-base pair can act as a buffer, resisting changes in pH when small amounts of acid or base are added to the solution.
In this system:
- The acetic acid is the acid (proton donor).
- The acetate ion is its conjugate base (proton acceptor).
These types of solutions are important in maintaining constant pH in biological systems (like blood, which has a buffer system made of carbonic acid and bicarbonate) and in many industrial and laboratory applications where pH control is required.